Advancing Precision in Clinical Research |Clival Database
Clival Database is a leading Clinical Trial Organization offering robust clinical trial data solutions tailored for the Pharma Clinical industry. As a comprehensive Clinical Organization, it supports every phase of research with advanced Clinical Statistical Analysis, ensuring accuracy and regulatory alignment. From data collection to outcome interpretation, Clival Database empowers sponsors and CROs with seamless integration and insights, accelerating innovation and improving the success rate of clinical trials worldwide. Explore smarter research with Clival Database.
Get More Info:- https://clival.com/cro

The Full Drug Development Process: What You Need to Know |Clival Database
Drug development is the step-by-step process of bringing a new medicine from the lab to the pharmacy. It includes drug discovery, lab research, testing in people, and approval by health agencies. Every step takes time and money. But it’s necessary to make sure medicines are safe and effective.
Get More Info:- https://www.studentinsta.com/r....ead-blog/4987_the-fu
Applying Complex Innovative Clinical Trial Designs to Orphan Drug Development
The design and development of orphan drugs, designed for rare diseases, are uniquely challenged as limited patient populations available for clinical trials. While more traditional designs may suffice in normal scenarios, there is a need for more complex innovative clinical trial designs (CIDs). CIDs design efficiently around advanced statistical methods and adaptive approaches to reduce trial timelines while maximizing drug safety for the few patients involved. This blog thus investigates how certain elements and strategic considerations tied to CIDs may be applied for orphan drugs.
Get More Info:- https://clival.com/blog/applyi....ng-complex-innovativ
Clinical Research Phases and the Path to Drug Approval
The process of developing a new drug is an intricate and multi-step endeavor that typically takes years to complete. The journey begins with drug discovery and development phases , which explore potential drug candidates, and then moves into clinical trials, regulatory reviews, and ultimately, market approval. Below, we’ll dive deeper into each of these key phases of drug discovery and development.
Get More Info:- https://clival.com/blog/clinic....al-research-phases-a
Clinical Research Phases and the Path to Drug Approval
The process of developing a new drug is an intricate and multi-step endeavor that typically takes years to complete. The journey begins with drug discovery and development phases , which explore potential drug candidates, and then moves into clinical trials, regulatory reviews, and ultimately, market approval. Below, we’ll dive deeper into each of these key phases of drug discovery and development.
Get More Info:- https://clival.com/blog/clinic....al-research-phases-a
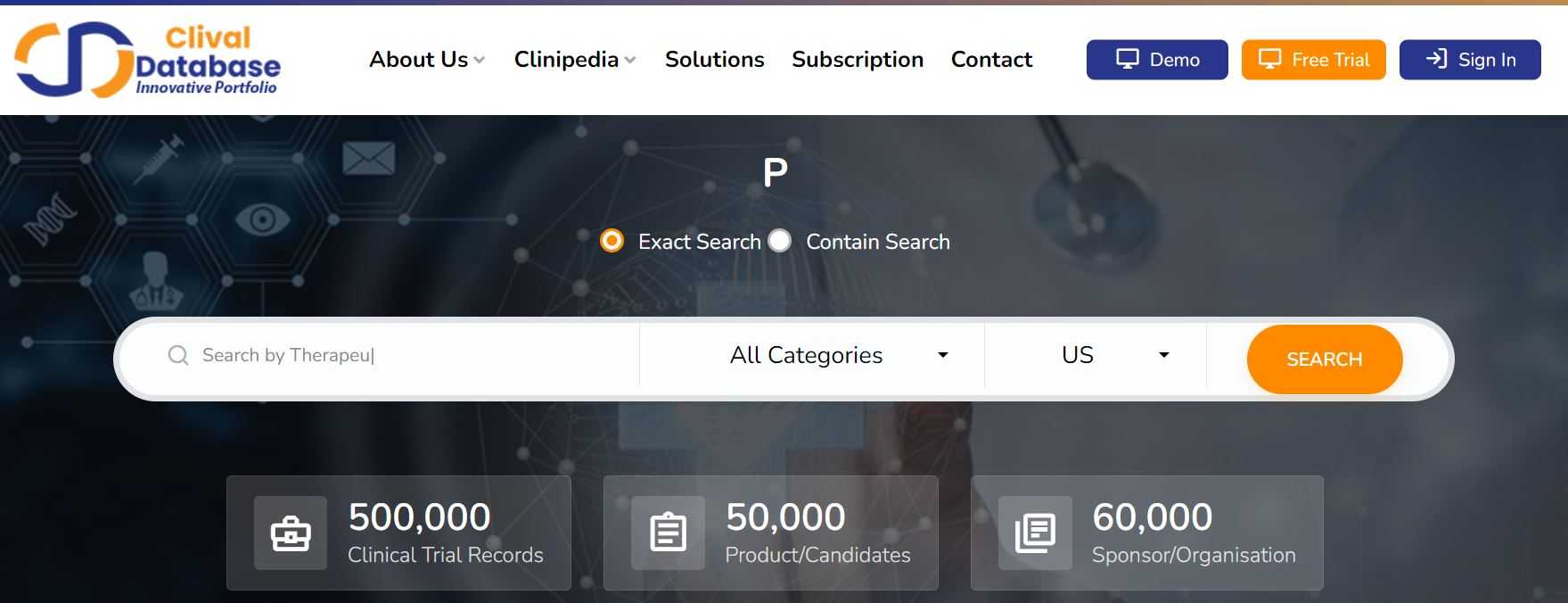
A clinical trials data platform offering insights on 500,000 clinical trials and innovative drugs for the pharmaceutical and allied industries.